 As we have discussed in previous posts, the care of patients with cardiac arrest is a key skill for Emergency Providers. ACLS provides a foundation for care but is rife with shortcomings including, but not limited to, reliance on outdated data and inability to adapt in the face of improved understanding of cardiac arrest pathophysiology. The incorporation of technological advances and skills is another massive limitation of ACLS. One of these technologies is point of care ultrasound (POCUS).
As we have discussed in previous posts, the care of patients with cardiac arrest is a key skill for Emergency Providers. ACLS provides a foundation for care but is rife with shortcomings including, but not limited to, reliance on outdated data and inability to adapt in the face of improved understanding of cardiac arrest pathophysiology. The incorporation of technological advances and skills is another massive limitation of ACLS. One of these technologies is point of care ultrasound (POCUS).
Over the last two decades, POCUS has become a integral part of Emergency Medicine training and practice. POCUS allows for rapid, bedside diagnosis of a number of conditions including cholecystitis, urinary retention and ectopic pregnancy. Additionally, it is becoming a greater component in the management of the critical patient where it can be used to assess cardiac contractility, wall motion abnormalities, intraperitoneal free fluid and more. Application of POCUS in all patients with cardiac arrest is simply the next step. This diagnostic modality is not highlighted in the current iteration of ACLS but is a practice changer. The bottom line is that application of POCUS in cardiac arrest allows the emergency provider to guide resuscitation with a direct look into the body – we are no longer blind.
For this post, I want to discuss two ways that we can use ultrasound in cardiac arrest patients, specifically in pulseless electrical activity (PEA), in the Emergency Department:
- Assessment for the presence or absence of cardiac output and
- As an alternate framework to the Hs and Ts.
A quick disclaimer – I am not an ultrasound expert, I did not do a fellowship but I am passionate about it’s application in our sickest patients.
ACLS Problem #1 – We assess for cardiac output by palpating for a peripheral or central pulse.
Palpating for a pulse with the small amount of real-estate and the end of our index and middle fingers has been the standard for hundreds of years. There are significant issues with using this method to assess for cardiac output:
- It is insensitive: Some patients with a cardiac output will not have a palpable pulse either because their blood pressure is too low, their body habitus makes it difficult to find the pulse, or because the providers finger tips are not sensitive enough. Additionally, the presence of gloves can make our sensation even more dulled.
- It isn’t specific: Providers may feel a pulse when one isn’t there. Typically, this occurs when a provider presses down hard over the vessel and confuses their own digital pulse for that of the patients.
- Inter-rater reliability of pulses is poor – Each of us has a different quality of sensation in our fingertips.
Tibballs and Russell demonstrated that rescuer pulse palpation was only 78% accurate (Tibballs 2008). Additionally, a study done 10 years prior demonstrated that providers had a difficult time rapidly (< 5 sec) locating the carotid pulse (Ochoa 1998). POCUS provides a superior tool to assessing for a cardiac output. Simply place the probe on the chest and look. Why trust your fingers (or someone else’s) when we can directly visualize the presence or absence of cardiac output. This is particularly relevant in patients who present with pulseless electrical activity (PEA). If chest compressions are already commenced, place the probe in the appropriate position while compressions are ongoing and look for cardiac activity during the standard pulse check. This ensures that cardiac images are obtained rapidly and that compressions aren’t held for prolonged periods.
In addition, we can swing the US probe down from the heart to the groin to find the femoral artery and guide placement of an arterial line. If we can’t palpate the femoral pulse, placement of an arterial line is extremely difficult and this technique is a rapid and easy way to improve success in placement.
Below is a video clip from a patient that was initially thought to be a PEA arrest. She had a narrow complex rhythm on the monitor but no palpable blood pressure as assessed by one doctor and one nurse.
Ultrasound clearly reveals organized activity. A femoral arterial line was placed revealing a blood pressure of 48/22. This isn’t cardiac arrest, it’s peri-arrest or massive shock. Instead of chest compressions and high-doses of epinephrine, which could significantly harm the patient, an epinephrine drip was started leading to improve blood pressure, cardiac output and mental status.

ACLS Problem #2 – ACLS stresses the “Hs + Ts” as a random array of possible PEA causes but does not provide a framework for management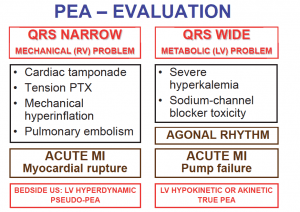
I’ve taught ACLS for years to providers in the hospital and have frequently been asked the question of how to use the “Hsand Ts” in actual patient management. Do we simply attempt to fix all of these issues on every patient with PEA? While many junior residents would be happy to place chest tubes for possible tension PTX, perform pericardiocentesis for possible tamponade and give thrombolytics for potential PEs, a more sensible approach would be to try and identify the actual cause.
Back in JUNE 2015, REBEL EM reviewed a new way to think about PEA arrest.
If we focus in on the “QRS Narrow” section of this algorithm, we can see how POCUS can be critical in rapidly determining the underlying pathology. A quick echo can reveal the presence of a pericardial effusion and assess for right atrial or right ventricular collapse establishing the diagnosis of cardiac tamponade.
Tension pneumothorax can be rapidly found with lung US looking for the absence of lung sliding.
We can organize our approach to US in the shocked (or “pulseless”) patient using the HI MAP mnemonic device: Heart, Inferior Vena Cava, Morrison’s Pouch, Aorta, Pneumothorax/Pipes. In addition to looking at the heart for cardiac tamponade, we can look for an acute dilation of the right ventricle (suggests pulmonary embolism) or a ruptured mitral valve (MI with valve rupture).
A quick look at Morrison’s Pouch can reveal occult bleeding from trauma or a ruptured ectopic pregnancy and we can swing over to the middle of the abdomen to look for a ruptured aortic abdominal aneurysm.
The bottom line is that ultrasound gives the provider a tool to diagnose reversible causes of arrest or peri-arrest. In a matter of minutes, we can rule in or out a variety of possible etiologies of cardiac arrest and severe shock (the prior categorized PEA arrest) and institute appropriate interventions (chest tube, pericardiocentesis, thrombolytics for PE) over blind administration of drugs (epinephrine) or interventions.

Future Directions
Where do we go from here? Application of POCUS to assess for cardiac output in lieu of the “pulse check” and diagnostic US looking for the cause of the arrest is well within the skill set of Emergency Physicians and is ready for incorporation now. A number of Emergency Physicians have been incorporating transesophogeal echo (TEE) into practice over the last 5-10 years. TEE offers a number of advantages in patients with cardiac arrest including better image resolution, ability to continually image during application of compressions (no need to pause) and allows for assessment of quality of compressions. Blaivas published an excellent case series highlighting the utility of TEE to guide management (Blaivas 2008). Again, TEE appears to be well within the skill set of Emergency Physicians and in the future, I think we’ll be utilizing this modality broadly in the critically ill patient. But that’s for another Beyond ACLS post.
Thank you to Marsia Vermeulen and Haney Mallemat for sharing the wonderful ultrasound images found in this post.
Read More
- Ultrasound Podcast: Ultrasound guided CPR Part 1. How we’re doing it wrong.
- Ultrasound Podcast: Ultrasound guided CPR Part 2. TEE & US = New pulse check
- Tibballs J, Russell P. Reliability of pulse palpation by healthcare personnel to diagnose paediatric cardiac arrest. Resuscitation 2009; 80: 61-4. PMID: 18992985
- Ohcoa FJ et al. Competence of health professionals to check the carotid pulse. Resuscitation 1998; 37: 173-5. PMID: 9715777
- Blaivas M. Transesophageal echocardiography during cardiopulmonary arrest in the emergency department. Resuscitation 2008; 78: 135-40. PMID: 18486300
Post Peer Reviewed By: Salim Rezaie (Twitter: @srrezaie)



