 Background: Despite serious concerns about the role of alteplase in the management of acute ischemic stroke including, but not limited to, significant conflicts of interest, unbalanced baseline patient characteristics, systematic devaluation of contrary data, lack of reproduced benefit and low fragility index, it remains standard care for patients presenting with symptoms of acute ischemic stroke within 3 (or 4.5 depending on system) hours of onset of symptoms. Though the NINDS studies only showed benefit in a specific subgroup of patients, subsequent work has endeavored to expand the target group in a classic example of indication creep. Patients with minor CVA (NIHSS < 5 without disabling features or, essentially mRS 0-1) represent one such subgroup in which alteplase is often not employed due mainly in part to the perception of minimal benefit with continued potential for harm (i.e. anaphylaxis, intracranial hemorrhage). Alteplase supporters argue that minor stroke patients should still get the drug as it not only may reduce symptoms but can also prevent deterioration. The evidence for this viewpoint is both extremely limited and of poor methodologic quality.
Background: Despite serious concerns about the role of alteplase in the management of acute ischemic stroke including, but not limited to, significant conflicts of interest, unbalanced baseline patient characteristics, systematic devaluation of contrary data, lack of reproduced benefit and low fragility index, it remains standard care for patients presenting with symptoms of acute ischemic stroke within 3 (or 4.5 depending on system) hours of onset of symptoms. Though the NINDS studies only showed benefit in a specific subgroup of patients, subsequent work has endeavored to expand the target group in a classic example of indication creep. Patients with minor CVA (NIHSS < 5 without disabling features or, essentially mRS 0-1) represent one such subgroup in which alteplase is often not employed due mainly in part to the perception of minimal benefit with continued potential for harm (i.e. anaphylaxis, intracranial hemorrhage). Alteplase supporters argue that minor stroke patients should still get the drug as it not only may reduce symptoms but can also prevent deterioration. The evidence for this viewpoint is both extremely limited and of poor methodologic quality.
Article: Khatri P et al. Effect of alteplase vs aspirin on functional outcome for patients with acute ischemic stroke and minor nondisabling neurologic deficits. The PRISMS randomized clinical trial. JAMA 2018; 320(2): 156-66. PMID: 29998337
Clinical Question: Does IV alteplase benefit patients with minor CVA (NIHSS < 5 without disabling features) when compared to aspirin.
Population: Adult patients (> 18 years of age) presenting with symptoms consistent with an acute ischemic stroke within 3 hours of onset. Only patients with an NIHSS < 5 and symptoms judged to be non-disabling were included.
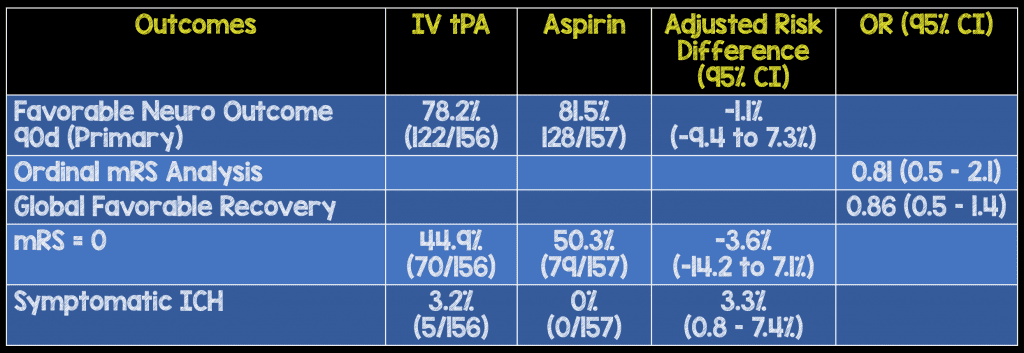
Outcomes:
- Primary: Risk difference in favorable neurologic outcome (defined as mRS 0-1) at 90 days.
- Secondary: Level of disability (assessed by mRS) and global favorable recovery at 90 days
Intervention: IV alteplase (0.9 mg/kg) + oral placebo
Control: Aspirin 325 mg PO + IV placebo
Design: Randomized, double-blind, double-placebo controlled trial
Excluded: Patients with significant prestroke disability (mRS = 2-6), dysphagia, intracranial hemorrhage on neuroimaging and other standard alteplase contraindications
Primary Results:
- 313 patients enrolled
- Aspirin arm: n = 157
- tPA arm: n = 156
- Mean age = 62
- Mean NIHSS score = 2
- Medain time of symptom onset to enrollment = 2.7 hours
Critical Findings:
Strengths:
- Asks a clinically important, patient centered question
- Only RCT to ask the question of whether alteplase improves outcomes in comparison to aspirin in mild stroke patients
- Randomization and blinding appropriately performed
- Baseline characteristics were well balanced
- 90% of patients completed the primary outcome
Limitations:
- The pharmaceutical manufacturer of alteplase was intimately involved in the trial introducing significant conflicts of interest
- The modified Rankin Scale (mRS) has been shown to have suboptimal inter-rater reliability. However, it is the standard accepted tool for neurologic outcome evaluation
- The study was stopped well short of the proposed enrollment number due to slow recruitment. The stoppage was initiated by the sponsor which raises concerns about access to data
- Selection bias: it was up to providers to determine if the deficits present were disabling or non-disabling
Discussion:
- The study was stopped early by the pharmaceutical company due to slow recruitment despite the researchers arguing for continuation. While the manuscript states that data had not been unlocked or unblinded at this point, the stoppage is suspicious. Had there been any kind of hint at improvement with alteplase, the study would likely have been completed as it would have been a powerful tool for the company to use in pushing for increased alteplase application. Early stoppage here suggests the sponsor may have been aware of the direction the data was pointing
- This is the only trial comparing alteplase to another active medication (aspirin) as opposed to simply comparing it to placebo.
- 3.3% of patients receiving alteplase had symptomatic ICH. In a group of patients with mild, non-disabling neurologic symptoms, any ICH rate is an unacceptable harm
- 13% of patients were found to have neurovascular mimics. 1 in 8 patients enrolled were exposed to a harmful medication with no hope for any benefit. This is concerning given that this is in an investigational setting (which likely performs better than in real life)
Authors Conclusions:
”Among patients with minor nondisabling acute ischemic stroke, treatment with alteplase vs aspirin did not increase the likelihood of favorable functional outcome at 90 days. However, the very early study termination precludes any definitive conclusions, and additional research may be warranted.”
Our Conclusions: Although the trial was stopped early, this methodologically sound RCT is the best evidence regarding the use of alteplase in mild, non-debilitating strokes. Given the absence of any other high-quality data and the absence of any hint of benefit to alteplase, onus must be squarely placed on the pro-alteplase group to prove a benefit to application of the drug in this group of patients.
Alteplase should not be routinely prescribed for patients presenting with mild (NIHSS < 5), non-disabling strokes until further research demonstrates benefit outweighing harm.
Potential to Impact Current Practice: Early stoppage of the study will prevent application of its findings. However, in an absence of any benefit (either in primary or secondary or exploratory outcomes) with a clear potential for harm, systems and providers should strongly consider withholding alteplase in this group until better evidence is available.
Bottom Line: There is no evidence that alteplase is beneficial in comparison to aspirin in improving neurologic outcomes in patients with mild, non-disabling strokes while there is clear evidence for harm in the form of symptomatic ICH.
References:
- Khatri P et al. Effect of alteplase vs aspirin on functional outcome for patients with acute ischemic stroke and minor nondisabling neurologic deficits. The PRISMS randomized clinical trial. JAMA 2018; 320(2): 156-66. PMID: 29998337
For More on This Topic Checkout:
- EM Lit of Note: Stopping the Alteplase Indication Creep
- EM Nerd: The Case of the Missing Comparator
- EMCrit: Podcast 116 – the tPA for Ischemic Stroke Debate
- PulmCrit: What is the Fragility Index of the NINDS Trial?
Post Peer Reviewed By: Salim R. Rezaie, MD (Twitter: @srrezaie)



