
- The original Sgarbossa criteria (i.e. the “weighted” Sgarbossa criteria) depends on a point system that rely on 3 findings, only 2 of which would provide enough points (i.e. 3) to make the diagnosis of AMI. Using the Sgarbossa criteria without the point system (i.e. the “unweighted” Sgarbossa criteria) increases sensitivity but decreases specificity.
- Sgarbossa et al diagnosed AMI by creatine kinase MB (CK-MB) elevations instead of angiographic evidence of acute coronary occlusion (ACO), which limits the sensitivity of the rule because it combines NSTEMI and STEMI patients in the outcome definition
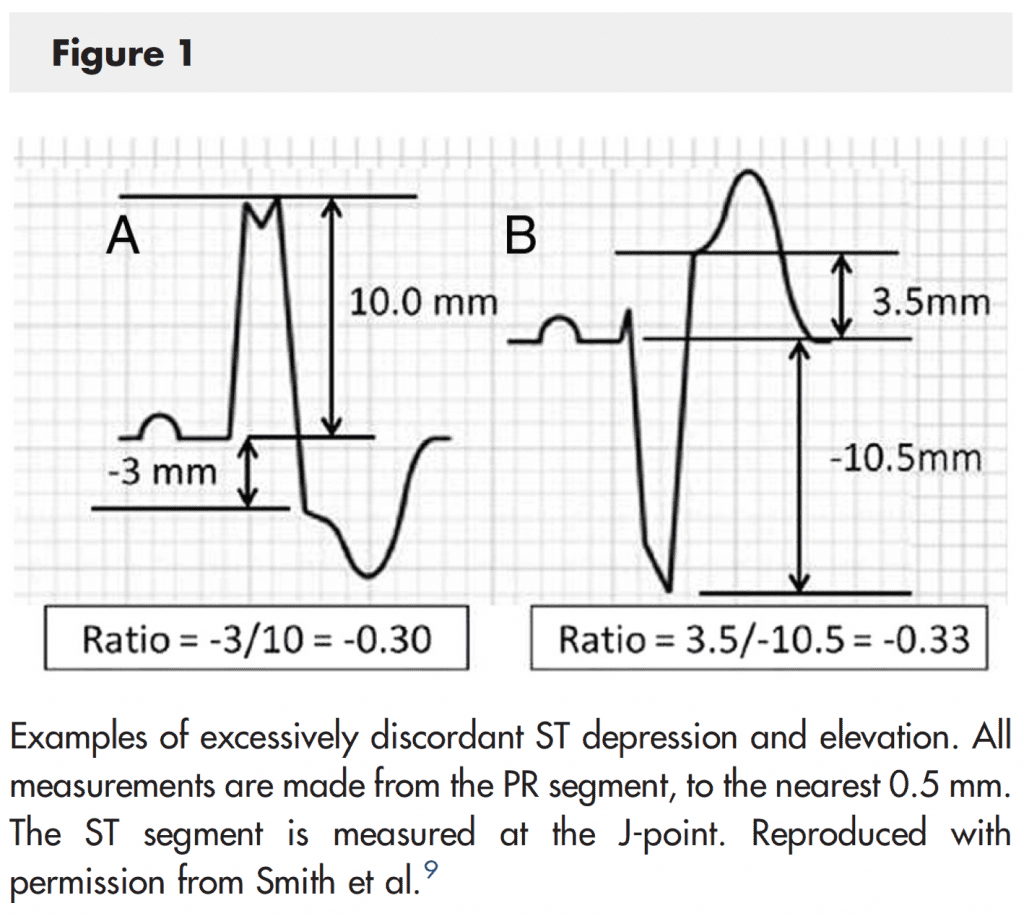
- Finally, Sgarbossa et al used an absolute criterion (5mm) rather than a proportional criterion for excessively discordant ST elevation lowering the sensitivity of the criteria.
The modified Sgarbossa criteria replaces the absolute 5mm discordant ST elevation with a proportion (ST elevation/S-wave amplitude ≤ -0.25). In other words, the modified Sgarbossa criteria only changes the last of the original Sgarbossa criteria with the first two criteria staying intact. Now, if any of these criteria are met, the cardiac catheterization lab should be activated. We have written on REBEL EM before about the modified Sgarbossa criteria and one of our conclusions was this rule looked very promising, but needed an external validation study. Well that study is now here and for full disclosure I am one of the authors on the paper.
What They Did:
- Multicenter, Retrospective, Case-Control Chart Review Trial
- Identification of LBBB and ACO defined as:
- Documented ACO on cardiac catheterization report
- Presumed Acute Occlusion with Significant Cardiac Outcome:
- Catheterization showing an acute but nonocclusive culprit lesion with very elevated cardiac biomarker (TnI ≥10 ng/mL, TnT ≥0 ng/mL, or CK-MB ≥80 ng/mL)
- If no angiography was done, then very elevated cardiac biomarker and a new or assumed new regional wall motion abnormality on cardiac echo
- ECG positive for any criteria with death before attempted emergent cardiac catheterization could be done
- ACO was determined by angiographic findings and cardiac biomarker levels
- Exclusion Criteria: hyperkalemia (>5.5 mEq/L), sever HTN (DBP > 120mmHg), extreme tachycardia (HR > 130 BPM), pulmonary edema with respiratory distress requiring mechanical ventilation, and cardiac arrest with no prearrest ECG available
Outcomes:
- Primary: Diagnostic accuracy of modified Sgarbossa criteria (in terms of sensitivity and specificity) vs the original Sgarbossa criteria for identification of ACO in patients with LBBB
Results:
- 45 patients with LBBB had ACO
- 249 patients with LBBB and no ACO were controls
- Modified Sgarbossa Criteria vs Orignal Weighted Sgarbossa Criteria:
- Sensitivity: 80% vs 49 % (p <0.001)
- Specificity: 99% vs 100% (p = 0.5)
- Modified Sgarbossa Criteria vs Unweighted Sgarbossa Criteria:
- Sensitivity: 80% vs 56% (p <0.001)
- Specificity: 99% vs 94% (p = 0.004)
Strengths:
- ACO was defined by cardiac catheterization findings and cardiac biomarkers, not just cardiac biomarkers alone
- Interobserver agreement for ECG measurements was 92% for QRS amplitude measurements within 1mm and ST deviation within 0.5mm
- Simple agreement and K value for ACO adjudication were 92% and 0.82, respectively
Limitations:
- Retrospective study, meaning some eligible patients could have been missed by the billing database
- There is a spectrum bias as this is a retrospective study. Not every patient with LBBB received a cardiac catheterization. In reality the group selected in this study is a sicker population than the general population of LBBB. This might actually strengthen and not weaken the results of the study as this is exactly the group that needed to be evaluated for ACO.
- ACO is a dynamic process, with coronary lesions known to occlude, reperfuse, and reocclude spontaneously. Using only angiographic occlusion as evidence for ACO, patients with an acute occlusion that spontaneously reperfused would not be captured in this study
- The modified Sgarbossa criteria have not been tested in a large sample of physicians without special interest in ECG interpretation. The addition of the proportionality criterion may result in decreased utility in general practice
Discussion:
- The modified Sgarbossa criterion performs similarly well if using a proportionality cutoff of -0.20 or -0.25. The -0.20 cutoff has a slightly higher sensitivity than the cutoff of -0.25 (84% vs 80%) and a slightly lower specificity 94% vs 99%). Therefore the decision to use -0.20 or -0.25 as the proportional discordance cutoff is not absolute but depends on pretest probability and clinical context.
Author Conclusion: “The modified Sgarbossa criteria were superior to the original criteria for identifying ACO in LBBB.”
Clinical Take Home Point: The modified Sgarbossa criteria is superior to the original Sgarbossa criteria in identification of ACO in LBBB, and it is reasonable to start incorporating it into practice at this time as a supplement to clinical judgment.
References:
- Meyers HP et al. Validation of the Modified Sgarbossa Criteria for Acute Cornoary Occlusion in the Setting of Left Bundle Branch Block: A Retrospective Case-Control Study. Am Heart J 2015; 170: 1255 – 1264. LINK HERE
For More on This Topic Checkout:
- The Breach: Introducing the Modified Sgarbossa Criteria
Post Peer Reviewed By: Anand Swaminathan (Twitter: @EMSwami)



