 Background: In 2016 the annual incidence of out-of-hospital cardiac arrest (OHCA) in the United States was roughly 360,000 and 209,000 for in-hospital cardiac arrest (IHCA) (Mozaffarian 2016). Though survival rates are relatively dismal, arrests in the setting of shock amenable rhythms – ventricular fibrillation (VF) and pulseless ventricular tachycardia (VT) – have an overall better rate of return of spontaneous circulation (ROSC). While cardiac defibrillation may be effective in terminating VF/VT, defibrillation does not prevent recurrence of VF/VT. According to the advanced cardiovascular life support (ACLS) algorithm, amiodarone is still the recommended first-line medication for shock refractory VF/VT. These recommendations stem from two hallmark studies which demonstrated improved ROSC and survival rates to admission after amiodarone was administered (Dorian 2002, Kudenchuk 1999). However, ROSC does not represent a clinically meaningful endpoint. Subsequent studies have questioned the utility of amiodarone in cardiac arrest from the perspective of improving survival to hospital discharge or survival with a good neurological outcome.
Background: In 2016 the annual incidence of out-of-hospital cardiac arrest (OHCA) in the United States was roughly 360,000 and 209,000 for in-hospital cardiac arrest (IHCA) (Mozaffarian 2016). Though survival rates are relatively dismal, arrests in the setting of shock amenable rhythms – ventricular fibrillation (VF) and pulseless ventricular tachycardia (VT) – have an overall better rate of return of spontaneous circulation (ROSC). While cardiac defibrillation may be effective in terminating VF/VT, defibrillation does not prevent recurrence of VF/VT. According to the advanced cardiovascular life support (ACLS) algorithm, amiodarone is still the recommended first-line medication for shock refractory VF/VT. These recommendations stem from two hallmark studies which demonstrated improved ROSC and survival rates to admission after amiodarone was administered (Dorian 2002, Kudenchuk 1999). However, ROSC does not represent a clinically meaningful endpoint. Subsequent studies have questioned the utility of amiodarone in cardiac arrest from the perspective of improving survival to hospital discharge or survival with a good neurological outcome.
Laina et al. Amiodarone and Cardiac Arrest: Systematic review and Meta-Analysis. Int J Cardiol. 2016; 221: 780-788. PMID: 27434349
Clinical Question:
- How does amiodarone affect return of spontaneous circulation, survival, and neurological outcome following cardiac arrest?
Methods:
- Studies had to include one of the following outcomes: ROSC, short-term survival (i.e. survival to hospital admission for OHCA and 24 hour survival for IHCA), survival to hospital discharge, and neurologic outcome, comparing amiodarone to placebo or other antidysrrhythmic drug (lidocaine or nifekalant)
- Studies selected via literature review of MEDLINE via PUBMED and the Cochrane Library. Used the search terms “cardiopulmonary resuscitation”, “cardio-pulmonary resuscitation”, “CPR”, “ventricular fibrillation”, “pulseless ventricular tachycardia”, “pulseless electrical activity”, “death, sudden”, “heart arrest, induced”, “amiodarone”, “cordarone”, “pacerone”, “nexterone” and “angoron” to find eligible studies. Included studies that were randomized or observational in nature of either adults or pediatric patients with either OHCA or IHCA.
Design: Systematic review and meta-analysis
Exclusion: All studies that did not meet inclusion listed above were excluded.
Data Extraction: Extracted data on study characteristics, participant characteristics, and the following outcomes: (1) ROSC (2) survival to hospital admission (3) survival to 24 hrs (4) survival to hospital discharge (5) neurological outcome.
Outcomes: Evaluated effects of amiodarone compared with alternative treatments (lidocaine, nifekalant, or placebo) on the following outcomes: (1) ROSC (2) survival to hospital admission (3) survival to 24 hrs (4) survival to hospital discharge (5) neurological outcome.
Results:
Study Characteristics
- Initially identified 1663 studies, of those studies 10 met inclusion criteria
- 4 randomized studies (3 double-blinded, 1 controlled)
- 6 retrospective studies
- 7 studies evaluated OHCA, 3 studies evaluated IHCA
- Total 5,326 patients (2,162 received amiodarone, 1422 received placebo, 1666 received lidocaine, and 76 received nifekalant)
- Individual study sample size ranged from 25 to 3,026 subjects
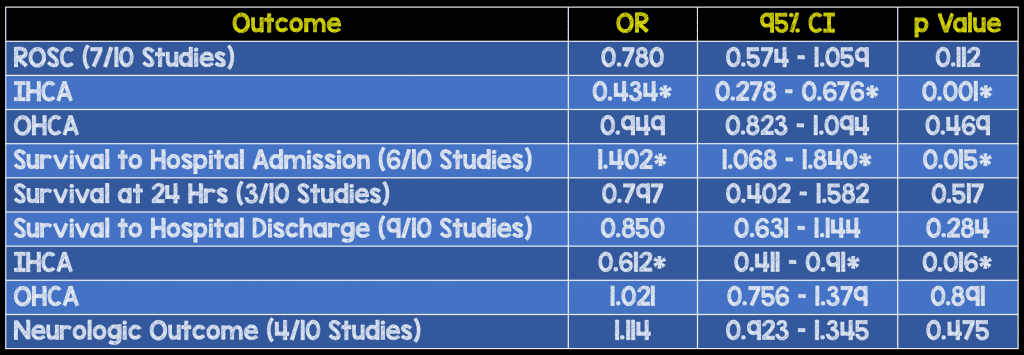
Critical Results:
Looked at effects of amiodarone versus alternative treatments (placebo or other antidysrhythmics) on the following 5 outcomes:
Strengths:
- Large overall sample size
- Includes 4 randomized control trials and 6 observational studies
- Looked at multiple outcomes including the patient relevant outcomes of survival to hospital discharge and neurologic outcome
- One of the only studies of its kind (i.e. systematic review/meta-analyses) that evaluate the effects of amiodarone on overall survival and favorable neurologic outcome
Limitations:
- Majority of studies (6 of 10) were retrospective. Given design of such studies, blinding was not possible leading to high risk of bias
- Only 3 included studies were adequately blinded and determined to have low risk of bias
- Limited studies looking at effects of amiodarone on both neurological outcome (4/10 studies) and survival at 24 hrs (3/10 studies)
- Unable to perform subgroup analysis on these two outcomes secondary to limited number of studies
Authors Conclusions:
“Amiodarone significantly improves survival to hospital admission. However, amiodarone does not improve survival to discharge or neurological outcomes compared to placebo or other antidysrhythmics.”
Our Conclusions: This systematic review and meta-analysis demonstrates that while amiodarone significantly increased survival to hospital to admission compared with placebo or other antidysrhythmics for OHCA, it does not significantly improve favorable neurological outcome or survival to hospital discharge. Ultimately, these outcomes are more meaningful from both a cost and quality of life perspective.
Potential to Impact Current Practice: Given this review and analysis, providers should consider removing amiodarone from their routine cardiac arrest algorithm and focus on interventions that have the ability to improve survival such as high quality chest compressions and defibrillation.
Clinical Bottom Line: Based on the available evidence, amiodarone does not appear to be associated with any meaningful clinical outcome in cardiac arrest including neurological outcome or survival to hospital discharge.
Guest Contributor

Alicia Skelton, MD
Bellevue/NYU Emergency Department
For More On This Topic Checkout:
- REBEL EM: Is Amiodarone Dead?
Amiodarone in Atrial Fibrillation:
- Core EM: Recent-Onset Atrial Fibrillation
- Core EM: Episode 98.0 – Cardioversion in Recent Onset AF
- Core EM: Ottawa Aggressive Atrial Fibrillation Protocol
Amiodarone in VT:
- Core EM: Procainamide vs Amiodarone in Stable Wide QRS Tachydysrhythmias (PROCAMIO)
- EM Nerd: The Case of the Dysrhythmic Heart
- REBEL Cast: PROCAMIO Trial
Amiodarone in Cardiac Arrest:
- Core EM: Amiodarone, Lidocaine or Placebo in OHCA
Post Peer Reviewed By: Salim R. Rezaie (Twitter: @srrezaie)



