 Background: Morphine is a commonly used medication in acute coronary syndromes (ACS) to help relieve pain which in turn can help reduce sympathetic tone. Over the past few years however, there has been some concern raised about the drug-drug interactions with antiplatelet agents causing impaired platelet inhibition as well as an association with worsened clinical outcomes. P2Y12 receptor antagonists (i.e. Clopidogrel, Pasugrel, Ticagrelor) are typically administered with aspirin (dual anti-platelet therapy) as one of the cornerstones of treatment for ACS. This drug-to-drug interaction can cause delayed inhibition of platelet activation and potentially worsen clinical outcomes.
Background: Morphine is a commonly used medication in acute coronary syndromes (ACS) to help relieve pain which in turn can help reduce sympathetic tone. Over the past few years however, there has been some concern raised about the drug-drug interactions with antiplatelet agents causing impaired platelet inhibition as well as an association with worsened clinical outcomes. P2Y12 receptor antagonists (i.e. Clopidogrel, Pasugrel, Ticagrelor) are typically administered with aspirin (dual anti-platelet therapy) as one of the cornerstones of treatment for ACS. This drug-to-drug interaction can cause delayed inhibition of platelet activation and potentially worsen clinical outcomes.
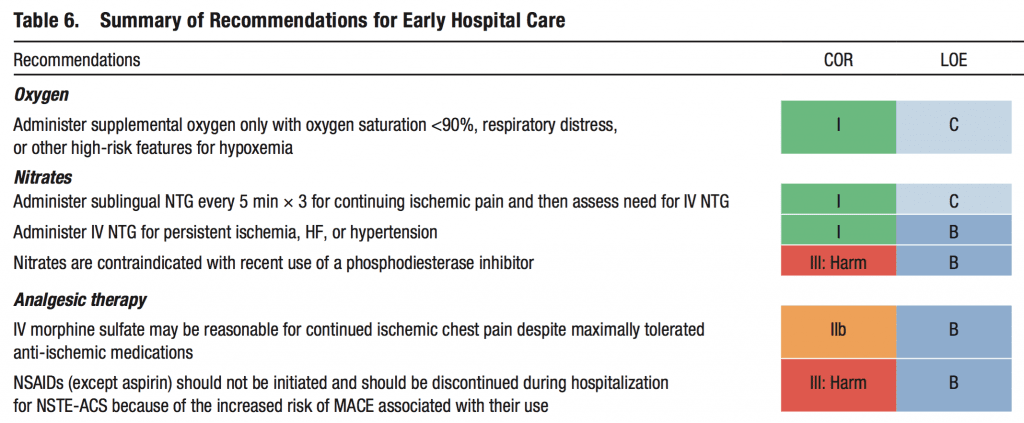
The last AHA/ACC guidelines for the management of patients with Non-ST-Elevation Acute Coronary Syndromes was published in 2014 [10]. There have been several trials published since its publication questioning several of the early treatment modalities commonly used in the pre-hospital and emergency department settings. The strength of recommendations is listed below:
Evidence that Morphine Inhibits P2Y12 Receptor Antagonists in Healthy Individuals
Study #1 [2]: Clopidogrel
- Randomized, double-blind, placebo controlled, cross-over trial
- 24 healthy subjects loaded with 500mg clopidogrel with IV placebo vs 5mg IV morphine
- Morphine delayed clopidogrel absorption, decreases plasma levels of clopidogrel active metabolite by 34%.
- Morphine also delayed maximal inhibition of platelet aggregation on average by 2 hours (2-fold increase from 1.25hrs to 3hrs) and in some cases up to 5 hours
- Bottom Line: Morphine slows down clopidogrel absorption, decreases plasma levels of its active metabolite, delays and diminishes the effects of clopidogrel on platelets in healthy volunteers.
Study #2 [3]: Ticagrelor
- Randomized, double-blind, placebo-controlled, crossover trial
- 24 healthy subjects loaded with 180mg ticagrelor with IV placebo vs 5mg IV morphine
- Morphine delayed ticagrelor absorption and its active metabolite by 1hr and decreased plasma levels of ticagrelor and its active metabolite by 25 – 31%
- Morphine did not retard or decrease ticagrelor antiplatelet effects
- Bottom Line: IV morphine decreases and delays ticagrelor effects, but this does not seem to decrease antiplatelet effects in healthy volunteers.
Study #3 [4]: Prasugrel
- Randomized, double-blind, placebo-controlled, cross-over trial
- 12 healthy volunteers loaded with 60mg of prasugrel with IV placebo vs 5mg IV morphine
- Morphine neither diminished total drug exposure nor significantly delayed drug absorption of prasugrel
- Morphine reduced maximal plasma concentrations of prasugrel active metabolite by 31% and delayed onset of maximal inhibition of platelet plug formation by 10 minutes
- Bottom Line: Morphine decreased maximal plasma concentrations of prasugrel and its active metabolite, but this did not seem to diminish the effects on platelets
There are several issues with each of these studies that need to be recognized:
- No patient oriented outcomes (These results are surrogate outcomes)
- Small studies (Largest study with 24 patients)
- Healthy patients, not ACS patients (Splanchnic circulation may be decreased in patients having ACS and therefore affecting absorption of medication)
Evidence that Morphine Inhibits P2Y12 Receptor Antagonists in STEMI Patients
Study #1 [5]:
- Prospective observation trial
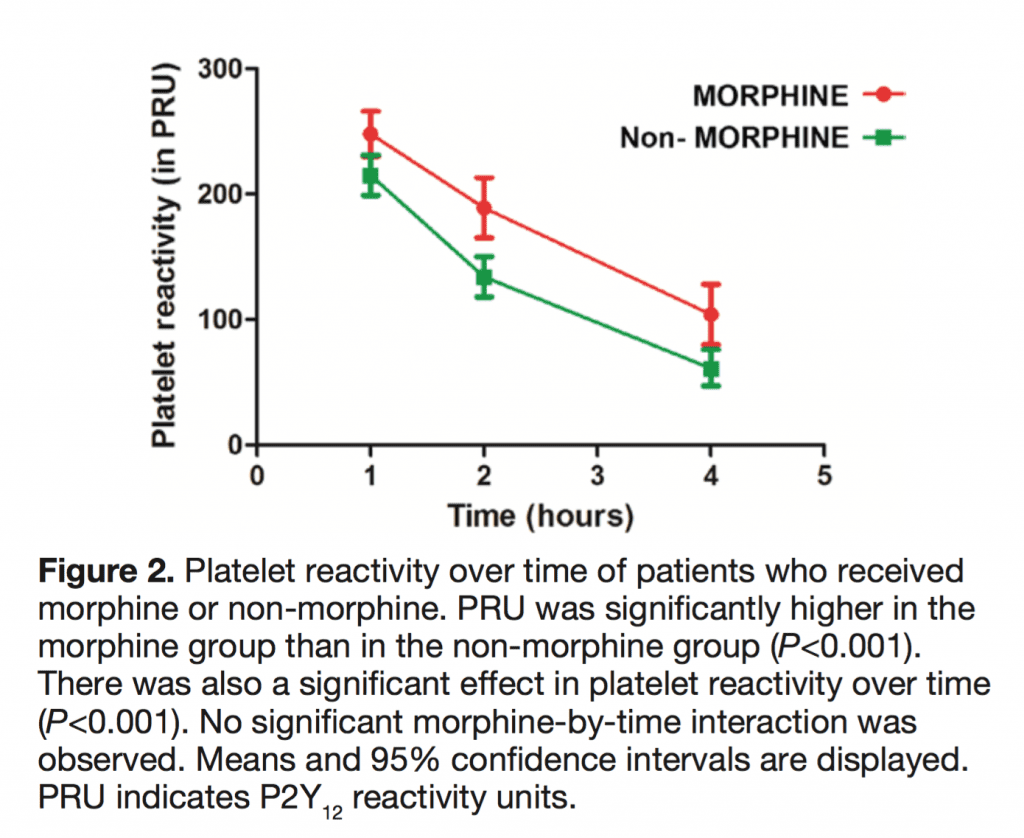
- 300 patients undergoing primary percutaneous coronary intervention for STEMI receiving prasugrel (n = 95) or ticagrelor (n= 205) had platelet reactivity assessed at 1, 2, and 4 hours
- Patients treated with morphine (n = 95 or 32% of patients)
- More vomiting: 15% vs 2%; p = 0.001
- Higher Platelet Reactivity Units 2 Hrs After Loading Dose: 187 vs 133
- Excluding Vomiting Patients Still with Higher Platelet Reactivity Units 2 Hrs After Loading Dose: 222 vs 107
- High Residual Platelet Reactivity (HRPR) at 2 Hours was Higher: 53% vs 29%
- In-Hospital Outcomes of Note (Morphine vs No Morphine):
- Death: 2% vs 3%; p = 0.53
- Reinfarction: 1% vs 1%; p = 0.58
- Stroke: 0% vs 1%; p = 0.33
- Dyspnea: 1% vs 8%; p = 0.10
- Bottom Line: In patients with STEMI, morphine use was associated with delayed onset of action of oral antiplatelet agents and increased platelet reactivity which could potentially lead to more thrombotic events
This study addressed two of the three issues brought up with the prior studies reviewed:
- Larger trial (300 patients)
- STEMI patients not healthy patients
- Still no patient oriented outcomes (These results are still surrogate outcomes)
Evidence that Morphine Use in ACS is Associated with Poor Clinical Outcomes [6]
What They Did:
- Evaluated the use of morphine within the first 24hrs after presentation in patients from the CRUSADE (Can Rapid Risk Stratification of Unstable Angina Patients Suppress Adverse Outcomes with Early Implementation of the ACC/AHA Guidelines) Quality Improvement Initiative
- Non-randomized, retrospective, observational registry of patients with Non-ST Elevation MI (NSTEMI)/ Acute Coronary Syndrome (ACS)
- Study population from 443 hospitals across the US (n = 57, 039)
- Outcomes evaluated in patients receiving:
- morphine versus no morphine
- morphine versus IV nitroglycerin
Outcomes:
- In-Hospital mortality
- Recurrent MI
- CHF
- Cardiogenic Shock
Inclusion:
- Patients included in the CRUSADE Initiative:
- Ischemic symptoms at rest within 24 hrs prior to presentation
- ST-segment depression ≥0.5mm
- Transient ST-segment elevation 0.5 – 1.0 mm (lasting for <10 min)
- Positive cardiac markers (elevated Troponin I or T)
Exclusion:
- Patients transferred from another institution
Results:
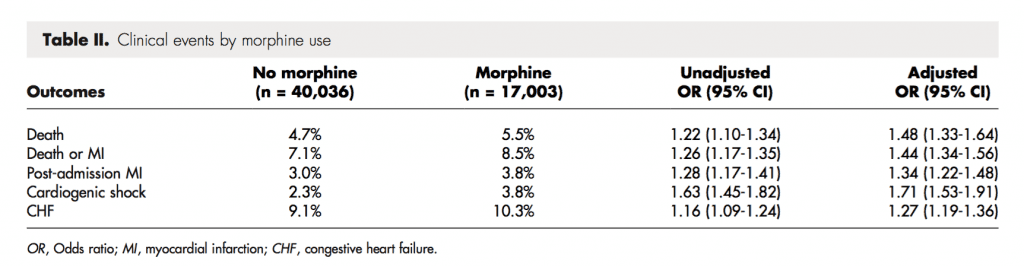
Patients receiving morphine within 24 hours of presentation: n = 17,003 (29.8%)
Clinical Events (Morphine vs No Morphine)
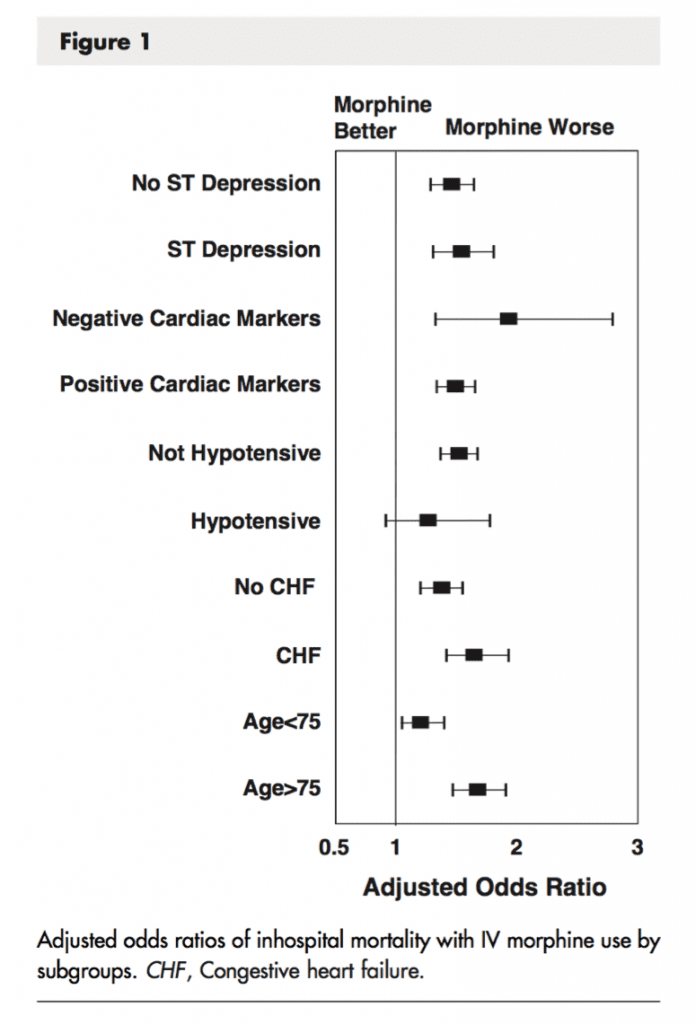
Subgroup Analysis (Morphine vs No Morphine)
Clinical Events (Morphine vs Nitroglycerin)
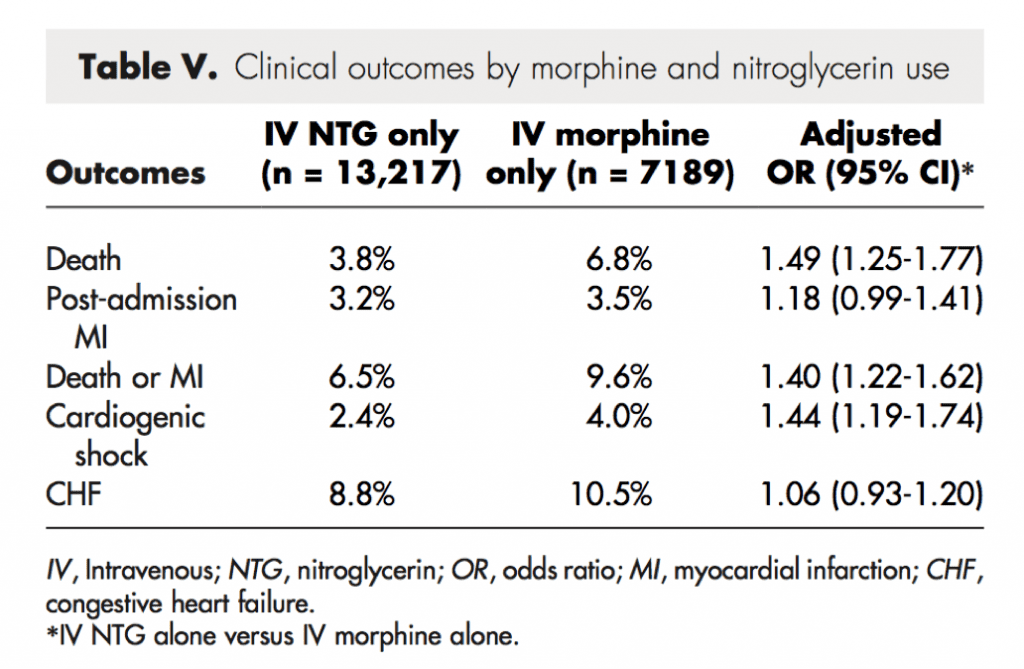
Strengths:
- Multicenter analysis
- Large cohort of patients
- Used propensity scoring to account for non-random treatment assignments
- Performed subgroup analyses to identify any subgroup variations in in-hospital mortality
Limitations:
- Observational trial not a randomized trial, therefore can only show associations not causation
- Patients receiving morphine were more likely to receive evidence-based medications and treatments than were patients who did not receive morphine. This difference in treatment was the exact opposite in the morphine vs nitroglycerin evaluation, with patients receiving morphine less likely to receive evidence-based medications and treatments
- Exact dosing and timing morphine and nitroglycerin were not collected in this database
- Non-fatal clinical outcomes were not rigorously defined
Discussion:
- There have been several other studies evaluating the association of morphine with adverse clinical outcomes with differing results:
- FAST-MI 2010 [7]: 2348 patients with STEMI; Morphine not associated with higher risk of clinical events, including death (Adjusted OR 0.48, 95% CI 0.12 – 1.85) and stent thrombosis (adjusted OR 1.31, 95% CI 0.36 – 4.74).
- Israeli 2008 Study [8]: 993 patients with STEMI/NSTEMI; Morphine not associated with higher 30 day mortality (OR 0.40, 95% CI 0.14 – 1.14)
Author Conclusion: “Use of morphine either alone or in combination with nitroglycerin for patients presenting with NSTE ACS was associated with higher mortality even after risk adjustment and matching on propensity score for treatment. This analysis raises concerns regarding the safety of using morphine in patients with NSTE ACS and emphasizes the need for a randomized trial.”
Our Conclusion: Given the observational, non-randomized nature of this study it is difficult to confidently conclude that morphine causes increase adverse outcomes in patients with NSTEMI/ACS, however the authors did a nice job with propensity scoring and subgroup analyses and the association of morphine with adverse outcomes should not be ignored.
Evidence for Fentanyl vs Morphine Use in ACS and Clinical Outcomes [9]
- Pre-hospital randomized double blind controlled trial
- 207 successive patients treated for suspected ischemic chest pain (187 included in the analysis)
- All patients received oxygen, aspirin, and nitroglycerin therapy
- If ischemic-type chest pain continued patients randomized to IV morphine vs IV fentanyl
- Primary Outcome: Incidence of Hypotension
- Secondary Outcome: Pain reduction
- Results:
- No statistical difference in pain reduction
- No statistical differences in hypotension (morphine 5.1% vs fentanyl 0%, p = 0.06)
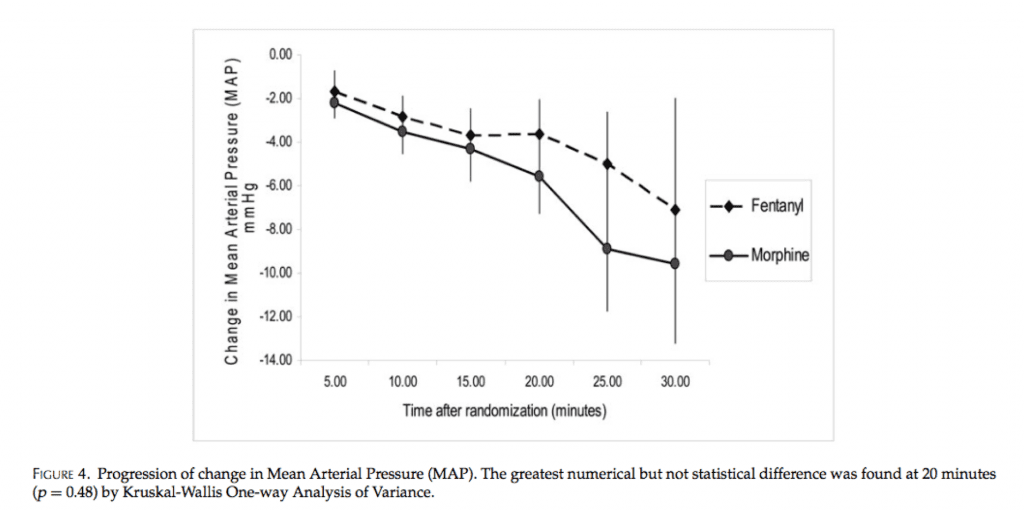
- Conclusion: Fentanyl and morphine were comparable in proving analgesia for ischemic-type chest pain with no statistical differences in hypotension
Clinical Take Home Point:
- The most robust evidence for the increase in adverse events and morphine use evaluates surrogate outcomes (i.e. Effect on platelet reactivity) in healthy volunteers. The findings in these studies should be read as hypothesis generating.
- When evaluating patient oriented outcomes, there are no randomized studies, but there are several registry level observational trials with differing results associating morphine with increased adverse event making it difficult to draw definitive conclusions.
- Until further stronger evidence is available it is probably worth erring on the side of safety and reserving morphine in an individualized manner evaluating benefit vs harm and use only when truly necessary in patients with refractory pain
- One possible alternative for refractory chest pain in ACS is fentanyl, although it would be nice to see studies evaluating the drug-drug interactions between fentanyl and P2Y12 antiplatelet agents
References:
- Giannopoulos G et al. P2Y12 Receptor Antagonists and Morphine: A Dangerous Liaison? Circ Cardiovasc Interv 2016. PMID: 27586412
- Hobl EL et al. Morphine Decreases Clopidogrel concentrations and Effects: A Randomized, Double-Blind, Placebo-Controlled Trial. J Am Coll Cardiol 2014. PMID: 24315907
- Hobl EL et al. Morphine Decreases Ticagrelor Concentrations but not its Antiplatelet Effects: A Randomized Trial in Healthy Volunteers. Eur J Clin Invest 2016. PMID: 26449338
- Hobl EL et al. Morphine Interaction with Prasugrel: A Double-Blind Cross-Over Trial in Healthy Volunteers. Clin Res Cardiol. 2016. PMCID: PMC4805697
- Parodi G et al. Morphine is Associated with a Delayed Activity of Oral Antiplatelet Agents in Patients with ST-Elevation Acute Myocardial Infarction Undergoing Primary Percutaneous Coronary Intervention. Circ Cardiovasc Interv. 2014. PMID: 25552565
- Meine TJ et al. Association of Intravenous Morphine Use and Outcomes in Acute Coronary Syndromes: Results from the CRUSADE Quality Improvement Initiative. Am Heart J 2005. PMID: 15976786
- Puymirat E et al. Correlates of Pre-Hospital Morphine Use in ST-Elevation Myocardial Infartion Patients and its Association with In-Hospital Outcomes and Long-Term Mortality: The FAST-MI (French Registry of Acute ST-Elevation and Non-ST-Elevation Myocardial Infarction) Programme. Eur Heart J 2016. PMID: 26578201
- Iakobishvili Z et al. Effect of Narcotic Treatment on Outcomes of Acute Coronary Syndromes. Am J Cardiol 2010. PMID: 20346305
- Weldon ER et al. Comparison of Fentanyl and Morphine in the Prehospital Treatment of Ischemic Type Chest Pain. Prehosp Emerg Care 2016. PMID: 26727338
- Amsterdam EA et al. 2014 AHA/ACC Guideline for the Mangement of Patients with Non-ST-Elevation Acute coronary Syndromes: A Report of the American college of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am coll Cardiol 2014. PMID: 25260718
For More Thoughts on This Topic Checkout:
- Ryan Radecki at EM Literature of Note: Yes, Let MONA Fade Away
- Rory Spiegel at EM NERD (EMCrit): The Case of the Inconsequential Truth
Post Peer Reviewed By: Anand Swaminathan (Twitter: @EMSwami)



